Arts & Sciences Research Paper #5: The most kindly way to preserve by Alesone Gray, Sugarwricht
Our fifth A&S Research Paper comes to us from Baroness Alesone Grey, Sugarwricht, and is drawn from her studies in historical confectionary, specifically the preserving of fruits with sugar. (Prospective future contributors, please check out our original Call for Papers.)
The Most Kindly Way To Preserve
One of the great joys of preserving the harvest is you can enjoy summer time fruits all year round, including in the dark days of winter. Many fruits have a very short season and need to be picked and eaten or preserved quickly. In the United States, the tart cherry season lasts 1-2 weeks in June. Apricots are 4-6 weeks starting around mid May. Peaches and plums start around the end of June and can last through August, depending on region. Apples, pears, quince and crab apples start their peak in September-October. There are a few varieties of fruits that have 2 seasons; among them are figs, which are picked in early spring and early fall.
But which method of preservation is ideal for various fruits? It depends.
Contents
The Science of Preservation
Some Historical Examples
Troubleshooting Preserves
The Science of Preservation
The choice of method depends on a number of factors including pectin, pH, acidity, ripeness and fragility of the fruit. These factors create a chemical equation, which dictate an ideal medium and agents for preserving fruits. The basic formula looks a lot like this:
Preserves = sugar + pectin + acid + fruit + heat
In an ideal world, both the gelling agent and the acid come from the fruit being processes. However, some fruits do not have enough natural pectin or acids to gel on their own. A preserve can be supplemented with a thickening agent like isinglass, if there is insufficient pectin in the fruit. If the fruit does not have enough acid to bind with the pectin efficiently, wine can be used to assist gelling.
In all of the preserve recipes we have from period, the key ingredient is sugar, in a ratio that is 1:1 equal parts sugar and other stuff (fruit, pulp, liquids). This ratio is significantly higher than most modern recipes.
Sugar raises the boiling point of water. This allows for more sugar to be dissolved into a solution, creating a super saturated concentration. This is important for development of jellies, conserves, preserves and syrups, as the final temperature determines texture and consistency. The chemical properties of sucrose include antioxidant properties, which helps prevent the deterioration of textures and flavor in preserved fruits. [1]
Sugar is also an antimicrobial agent that works via osmosis and diffusion. Osmosis is the process of establishing equilibrium by drawing water into super-saturated sugar water from inside the fruit. This reduces the number of free water molecules in the food; these are needed for microbial growth. This is a state of dehydration for the microbes and they cannot multiply and cause spoilage. When equilibrium has been achieved, the fruit will enrich itself with sugar solutes in the syrup solution via diffusion.[2]
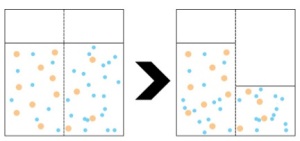
Pectin is the second ingredient that is vital to a cohesive preserve. Pectin was originally identified as a substance in 1790 by French chemist Nicholas Vauquelin.[3] The pectin source was an apple.
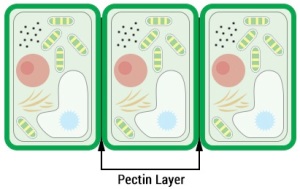
Pectin is a complex carbohydrate. It is located between the cell walls of plants, binding the cells together by creating a continuous layer between adjacent cells. In confections, pectin is a gelling agent, thickening agent and stabilizer.
Fruit that has just become ripe contains the most amount of pectin. As fruit continues to ripen, pectin is broken down and loses its ability to be a gelling agent. Ripening turns pectin into pectic acid.[4] This creates a fruit with a full bodied flavor, but little pectin. These fruits are great for eating, but not very good for preserves.
| Fruits high in pectin | Fruits low in pectin |
| Pears Apples Quince Plum Gooseberries Oranges Lemons Limes Currants Crab apples Blackberries |
Cherries Grapes Raspberries Strawberries Apricots Peaches |
Pectin cannot function on its own. It needs sugar and an acid in order to bind the preserve together. Sometimes there is sufficient acid in the fruit that a gentle application of heat is enough to create the bond. Often times there is not and fruit requires from an additional acid. In period recipes this acid is often wine.
It is important to pair the right wine with the right fruit. Acids in wine develop as the grape matures. In warmer climates, acids are lost through plant respiration. Warmer weather results in lower acidity, and cooler weather creates higher acidity. A wine produced in Napa, California will have a lower natural acidity than a wine produced in Chablis, France.
When looking for a wine to use, find one with a low pH; the lower the pH number, the more acidic the variety. Most table wines have a pH somewhere between 3.3 and 3.7;[5] white wines tend to be more acidic than their red counterparts. Regions that produce fairly acidic wines include: Chablis (France), Austria, Germany, and Northern Italy for white wines and Bordeaux and Burgundy in Southern France for red wines.
Occasionally a fruit both low in acid and low in pectin can use the assistance of a 3rd party gelling agent. In period this could come in the form of isinglass. Isinglass is a type of gelatin that comes from the air bladders of fish. Typically this would be sourced from sturgeon and would be sold dried, either whole or shredded. Isinglass is an ideal gelatin as it is odorless and tasteless. This means it will not impart additional flavor to the finished preserve. Unlike a modern gelatin, it does not need to bloom (become fully softened and melted) to be at full strength, and can added directly to the preserve as it nears completion of cooking. It completely dissolves and will cause some opaqueness to the finished product. Unlike a jelly using modern gelatin, when it sets, it will not be clear.

The amount of isinglass used will determine the setting properties of the finished product. A typical recipe can use 1-3 oz of isinglass. Modernly isinglass comes from cod. It can be found with brewing supplies, as it is a clarifying agent for beers and wines. However, it is advisable to stay away from liquid and powdered isinglass as they often had added citric acid and other materials for extending shelf life. To get the best results use whole or shredded isinglass.
The final factor in making preserves is heat. How do you know when the preserves are done, and why? 220 degrees Fahrenheit is the magic number for preserves, 8 degrees above a boil. Sugar at this stage is roughly 78% concentrated. It is also the temperature where sugars and pectin form a gel bond.
To test for this stage, you can do several things.
- Use a candy thermometer. 220 degrees should be clearly marked.
- When the temperature reaches 220 degrees, dip a spoon into the rolling mixture and watch what happens as the liquid runs off. If the liquid drops that form together into a sheet off of the spoon, the gel point has been reached. If the liquid retains droplet form, then it needs a little more time.
- You also can test your gel on a frozen plate by dropping a small bit of gel on a plate that has been chilling in the freezer. If the mixture gels then it is done.
- The period method would be a combination of what the bubbles look like as the mixture is boiling and the spoon sheeting test. Bubbles will be well formed, but not rolling. They will still pop like bubbles.
Back to Top
Some Historical Examples
Preserves with additional acids
“To make conserve of any fruit you please, you shall take the fruit you intend to make conserve of and if it be stone fruit you shall take out the stones; if other fruit, take away the paring and core, and them boil them in fair running water to a reasonable height; then drain them from thence, and put them into a fresh vessel with claret wine, or white wine, according to the color of the fruit; and so boil them to a thick pap all to mashing, breaking, and stirring them together; then to every pound of pap put to a pound of sugar, and so stir them all well together, and being very hot, strain them through fair strainers, and so pot it up.” [6]

In this recipe by Markham (1615), the author is supplementing potential pectin levels with additional acids. He is calling for a 1:1 ratio of sugar to everything else. This creates a super sugar solution. Fruits cooked with this method have a higher potential of becoming candy-like than a modern preserve. The wine adds a fantastic level of flavor to the over all preserve. It also helps the pectin bind better to create a more semi-solid gel state. Cherries using this recipe with a red wine end up with a consistency that is similar to a very thick molasses.
This is a great recipe for stone fruits with lower pectin levels like cherries, apricots and peaches. Cherries set well with a nice red from the Bordeaux region. Apricots and peaches set well with a nice German Riesling. I prefer not to use this recipe with high pectin fruits such as currants and gooseberries. I have found the gels set up too fast and are thick with a gritty texture.
Preserves with additional gelling agent
“To make jelly of Straw-berries, Mulberries, Raspberries, or any such tender fruit. Take your berries, and grind them in an Alabaster mortar, with four ounces of sugar and a quarter pint of fair water, and as much Rosewater: and so boil it in a possinet with a little piece of isinglass, and so let it run through a fine cloth into your boxes, and so you may keep it all the year.” [7]
In this recipe from Hugh Plat from 1609, we have several factors working against us. The sugar to water/pulp ratio is low. We are not creating a super sugar concentration as the liquids outweigh the sugar. We are working with fruits that are naturally lower in pectin. And we are not adding any additional acids to help our pectin. In this case the author suggests adding isinglass as a 3rd party gelling agent to thicken soft fruits low in pectin.
We also are not given an indication of how long it is to be cooked. I have found I get the best results when I put isinglass in around the 218 degree mark. It will dissolve rapidly. Your preserve will continue to cook a bit as it rises into the 220 degree range. You can add more isinglass at this stage if you want to and the mixture is not gelling to your liking. If it rises beyond 224 and begins heading toward the stage where it will form a soft ball in cold water, you have gone too far. Unfortunately, there are a lot of external factors that make executing this recipe difficult. However the end result should be an opaque gel with a very intense flavor profile.
Preserve with supplemental pectin
To make Marmalade of Lemons or Oranges. Take ten lemons or oranges and boil them with half a dozen pippins, and so draw them through a strainer, then take so much sugar as the pulp does weigh, and boil it as you do Marmalade of Quinces, and then box it up.
In this recipe, Plat is supplementing the citrus with pectin in the form of boiled pippins. This is another super saturated sugar concentration, i.e., it starts at 1:1 and concentrates further with cooking. The additional apple pectin creates a finished pulp that has a thick body that can be cut when it sets up.
Back to Top
Troubleshooting preserves
Food science is never 100% clear-cut, “follow this recipe and everything will work perfectly”. There are so many different elements that can cause a recipe to fail. When making fruit preserves, usually this manifests by your preserves not setting.
Possible variables
- Time
- Batch size
- Pectin
- Acid
- Sugar
- Ripeness
- Humidity
- Altitude
- Heat
- Cookware
- Canning procedures
It’s a pretty long list. Here are the four biggest variables to consider: all things being equal, what could I have done differently?
- How high was your heat?
- Pectin will break down at high temperatures. If you crank the heat to high and set your preserves to just do their thing, you will get through sugar stages fairly fast, but you can also break down your pectin. You will end up with very lovely syrup.
- To fix this problem: you can add water and re-saturate your sugar syrup and bring it back up to temperature via a lower temperature. You can also try adding a 3rd party gelling agent, like isinglass. You can also leave it in syrup state.
- Was your fruit too ripe?
- Just ripe fruit has the most natural pectin.
- To counter this, mix fruit that is too ripe with fruits that are either just ripe or slightly under ripe.
- Did you need to use an acid?
- Lower pectin fruits need the acid to help them.
- Unfortunately, this doesn’t really have a fix outside of re-saturating and adding a third party agent. You may find you need to use a lot more isinglass to get it to set up.
- Would more pectin help?
- You can always attempt the recipe and add a few apples. This is especially useful for fruits that have a lot of liquid in the cooking process.
Corrective measures should occur once your preserves have cooled completely. If you are using a modern canning water bath process you will be able to see in a few days whether they have set up. If after a few days the preserves have gelled to your liking, you can pop the lids and attempt a corrective method. It is always an option to leave it in a syrup state. Syrups are a perfectly period way to preserve foods.
Your ingredients will always play a large role in preserves. If you are using a sugar that is “in the raw”, “minimally processed” or has been crafted and cured in small batches, you will have additional preserve challenges. These sugars often have impurities that will need to be removed during the cooking process. They require longer times and reduced heat cooking temperatures. To rush the process can cause the pectin to break down faster and you will end up with lovely syrups.
When experimenting with preserves, I recommend trying a variety of cooking techniques with your fruits. You will discover which flavor combinations you like best. There is nothing to say you cannot cook a fruit using any of the period recipes, as long as you understand the properties of your fruits.
Back to Top
[1] “The Canadian Sugar Institute.” Functional Properties of Sugar. Accessed 07 Oct. 2015.
[2] “How Do Salt and Sugar Prevent Microbial Spoilage?” Scientific American Global RSS. Accessed 07 Oct. 2015.
[3] “Introduction to Pectin.” CyberColloids, Ltd. Accessed 08 Oct. 2015.
[4] “Science of Cooking: Pectin and Partners for Perfect Preserves.” Exploratorium: The Museum of Science, Art and Human Perception. Accessed 08 Oct. 2015.
[5] The Oxford Companion to Wine. Ed. Jancis Robinson and Julia Harding. Oxford: Oxford University Press, 2015. 549.
[6] Markham, Gervase, and Michael R. Best. The English Housewife: Containing the Inward and Outward Virtues Which Ought to Be in a Complete Woman … Montreal: McGill-Queen’s University, 1998.
[7] Plat, Hugh. “The Arte Of Preserving Conserving, Candying.” Delights for Ladies: To Adorne Their Persons, Tables, Closets, and Distillatories with Beauties, Banquets, Perfumes, and Waters. Reade, Practise, and Censure. London: Humfrey Lownes, 1609.
